Pharmaceutical Project Engineering and Construction Package From A to Z
Leader for IV solution turnkey projects supplier. Complete facilities to produce IV Fluids and Parenteral Solutions in Large (LVP) volumes with capacities from 1500 up to 240,000 pcs/h.
Pioneer supplier providing integrated engineering solutions in compliance with EU GMP, US FDA cGMP, PICS, and WHO GMP.
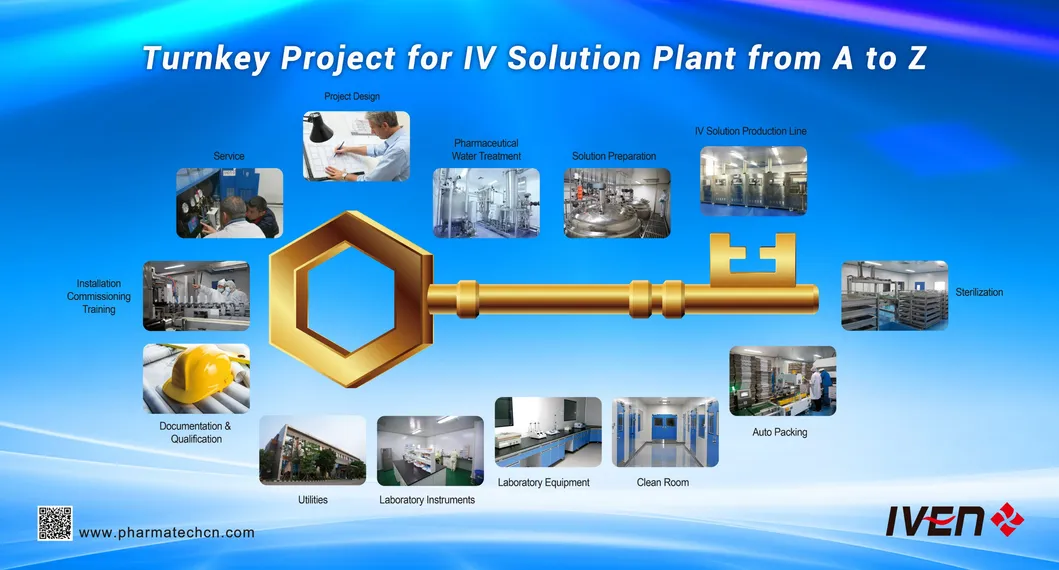
⚙️ Core Capabilities
Turnkey projects including localized procurement assistance and advanced know-how transfer:
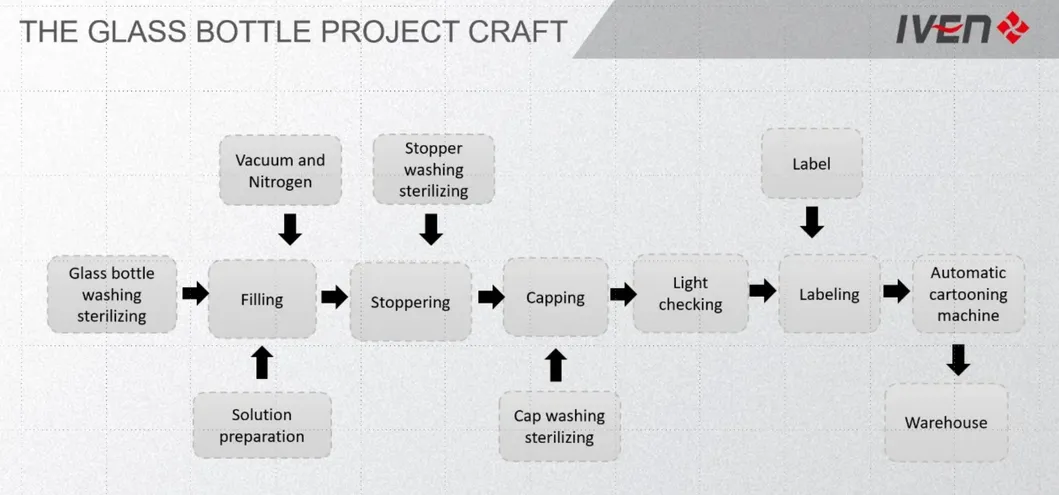
- Advanced Know How Transfer for Glass bottle IV solutions
- Post-Start up assistance
- Raw Materials and Consumables
- Black Utilities
Standard IV Solution Products & TPN
- NaCl 0,18 - 2.7% / Glucose 2,5 - 50%
- Sodium Lactate (Hartmanns's) / Ringer Lactate
- Water For injection / Sterile Water for Irrigation
- TPN (Total Parenteral Nutrition): Amino acids, Fat Emulsion
- Special Solutions: Paracetamol, Metronidazole, Ciprofloxacin
✅ Validation & Compliance
The standards of good manufacturing practice (cGMP) require special attention to risk assessment and verification procedures. We provide comprehensive Validation Master Plans ensuring all equipment and procedures meet quality integrity standards.

Turnkey Steps & Overseas Projects
√ Basic & Detailed Engineering √ HVAC and air treatment √ Clean Rooms √ Pharmaceutical Water Systems √ Training √ IQ/OQ/PQ Protocols √ Standard Operating Procedures
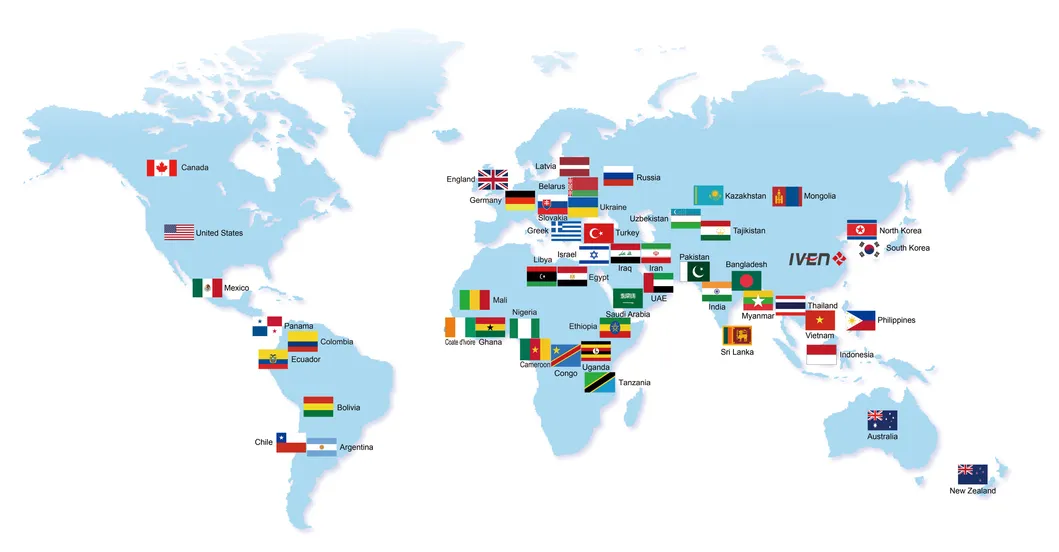

Company Profile







Frequently Asked Questions
Q1: What types of IV solutions can your production lines handle?
A: Our lines are versatile and can produce Non-PVC soft bag, PP bottle, and Glass vial IV solutions, as well as TPN, electrolytes, and special solutions like Paracetamol or Metronidazole.
Q2: Do your projects comply with international GMP standards?
A: Yes, all our pharmaceutical turnkey projects are designed and constructed in strict compliance with EU GMP, US FDA cGMP, PICS, and WHO GMP standards.
Q3: What is included in your "Turnkey" service package?
A: Our A to Z package includes basic/detailed engineering, clean room construction, HVAC systems, equipment installation, staff training, and full validation (IQ/OQ/PQ protocols).
Q4: What is the typical production capacity of the IV fluid lines?
A: We offer flexible capacities ranging from 1,500 up to 24,000 pieces per hour, depending on the specific product type and customer requirements.
Q5: Do you provide support after the factory starts operation?
A: Yes, we provide post-startup assistance, initial know-how transfer, and can supply raw materials, consumables, and spare parts for several years of operation.
Q6: In which countries have you successfully implemented turnkey projects?
A: We have delivered hundreds of sets of equipment and turnkey plants to over 40 countries, including Russia, Indonesia, Thailand, Saudi Arabia, Nigeria, and even Germany.



































 Premir Plastic Package
Premir Plastic Package